What is Battery Internal Resistance?

What is Battery Internal Resistance?
Battery internal resistance is the resistance within a battery that arises from the electrochemical processes and materials inside the battery. This resistance determines how easily current can flow within the battery, directly impacting the battery's performance.
- 1. Voltage Drop Under Load Method:
- 2. Impedance Spectroscopy:
- Why is it Important?
How is Internal Resistance Calculated?
There are typically two methods used to calculate battery internal resistance:
• A specific load (resistor) is connected to the battery, and a certain current is drawn from it.
• The open-circuit voltage 𝑉0 (before the load is connected) and the voltage under load 𝑉 are measured.
• If the current drawn from the battery 𝐼 is known, the internal resistance 𝑅𝑖 can be calculated using the following formula:
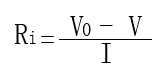
In this formula, 𝑉0 − 𝑉 represents the voltage drop, and 𝐼 represents the current drawn.
• This method measures the battery's internal resistance by applying small AC signals at different frequencies. It is a more complex method and is typically used in laboratory settings.
• Efficiency: Low internal resistance increases the efficiency of energy storage and transfer within the battery. High internal resistance can lead to energy loss and cause the battery to heat up.
• Performance: A battery with high internal resistance will experience more voltage drop under high current loads, which can negatively affect the performance of devices.
• Lifespan: High internal resistance can shorten the battery's lifespan. As internal resistance increases over time, the amount of energy the battery can deliver decreases.
• Safety: If the internal resistance is too high, the battery may overheat during use, leading to potentially dangerous situations.
In summary, the internal resistance of a battery is a critical parameter for both the overall performance and safety of the battery. Therefore, battery manufacturers and users aim to keep internal resistance as low as possible.

